Why the United States Next-Generation Sequencing Market Is Heading Toward a Genomic Revolution
From precision medicine and cancer diagnostics to drug discovery and rare disease testing, next-generation sequencing is rapidly transforming healthcare in the United States.
Why the United States Next-Generation Sequencing Market Is Heading Toward a Genomic Revolution
Healthcare in the United States is changing quickly, and one of the biggest reasons is the rise of Next-Generation Sequencing (NGS). Once considered a highly specialized research tool, NGS is now becoming an essential part of modern medicine. From identifying cancer mutations to diagnosing rare inherited conditions and accelerating drug discovery, this technology is reshaping how doctors, scientists, and pharmaceutical companies understand disease.
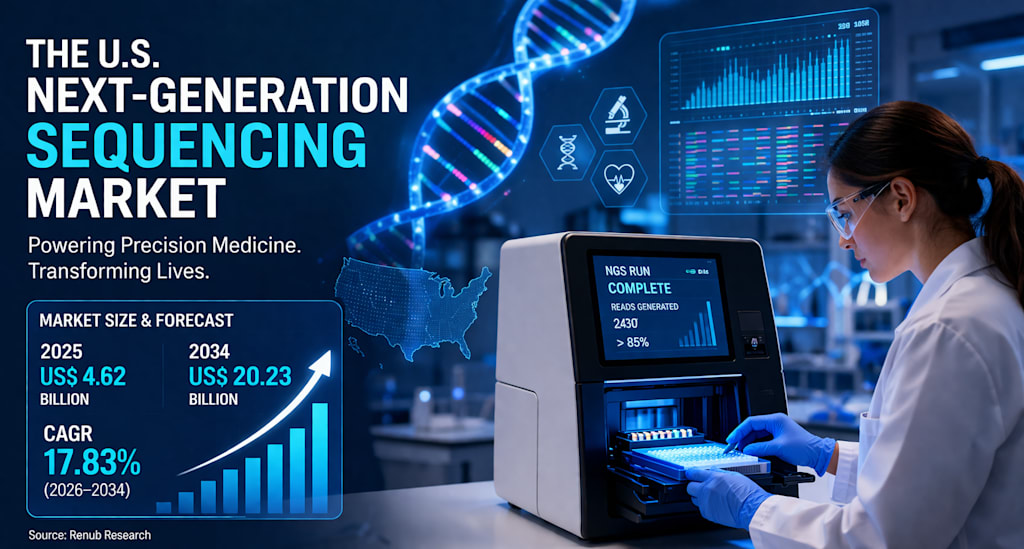
According to the market data you provided, the United States Next-Generation Sequencing Market is expected to grow from US$ 4.62 billion in 2025 to US$ 20.23 billion by 2034, expanding at a compound annual growth rate (CAGR) of 17.83% from 2026 to 2034. That kind of growth reflects more than just business momentum — it signals a much deeper transformation in the future of healthcare and life sciences in America.
The story of this market is not simply about machines and data. It is about a healthcare system moving away from one-size-fits-all treatment and toward more precise, personalized, and predictive care.
What Is Next-Generation Sequencing and Why Does It Matter?
At its core, Next-Generation Sequencing is a powerful genomic technology that can analyze millions of DNA or RNA fragments simultaneously. Compared to older sequencing methods, NGS is faster, more scalable, and far more efficient. It can provide detailed insights into genes, mutations, expression patterns, and molecular abnormalities that would otherwise remain hidden.
That matters because healthcare is increasingly becoming data-driven and individualized. A tumor in one patient may behave very differently from the same tumor type in another patient. A child with a rare genetic condition may spend years without an accurate diagnosis unless advanced sequencing is used. A pharmaceutical company may waste time and money developing a drug unless it understands the genetic pathways involved in a disease.
NGS helps solve these problems.
It is already widely used in:
Cancer genomics
Rare disease diagnosis
Infectious disease research
Prenatal and fertility testing
Drug development
Personalized medicine and companion diagnostics
This broad utility is one reason the U.S. has become one of the most important markets for genomic sequencing in the world.
Precision Medicine Is Fueling the Market
One of the strongest growth engines behind the U.S. NGS market is the rise of precision medicine. In simple terms, precision medicine means tailoring treatment to a patient’s unique genetic makeup, biology, and disease profile rather than relying only on general treatment guidelines.
This shift is especially important in oncology. Cancer treatment is no longer based solely on where a tumor is located in the body. Increasingly, it depends on which genetic mutations are driving that cancer. NGS makes it possible to identify those mutations quickly and accurately, helping physicians choose targeted therapies that are more likely to work.
The same logic applies to inherited disorders, neurological diseases, cardiovascular conditions, and even some infectious diseases. The more clinicians rely on molecular data, the more sequencing becomes a necessary part of care delivery.
This trend is not temporary. It is structural. Hospitals, biotech firms, pharmaceutical companies, and diagnostic laboratories are all investing in genomic workflows because the future of medicine is increasingly centered on molecular-level decision-making.
The U.S. Has a Major Structural Advantage
Another reason the American NGS market is growing so quickly is that the country already has a powerful ecosystem to support it.
The United States has:
World-class research universities
Advanced hospital systems
Strong biotechnology clusters
Deep pharmaceutical R&D pipelines
High public and private investment in genomics
That combination creates an ideal environment for sequencing adoption.
Research institutions continue to use NGS in cancer biology, functional genomics, infectious disease surveillance, and translational medicine. At the same time, private companies are developing faster instruments, more reliable reagents, and more sophisticated bioinformatics tools.
In other words, the market is being supported from both ends — research and commercialization.
That matters because some healthcare technologies fail when they cannot move from the lab into real-world use. NGS is doing the opposite. It is steadily moving from specialized research settings into hospitals, clinical labs, and mainstream healthcare systems.
Technology Improvements Are Making NGS More Accessible
One of the biggest reasons for NGS expansion is that the technology itself keeps improving.
Over the past several years, sequencing platforms have become:
Faster
More accurate
Easier to automate
More affordable
Better integrated with data analysis tools
This is a critical point. Advanced healthcare tools often struggle because they are too expensive or too difficult to use at scale. NGS is gradually overcoming those barriers.
Compact bench-top sequencers are making it easier for hospitals and smaller laboratories to adopt the technology without needing a massive infrastructure investment. Improvements in sequencing chemistry and workflow automation are reducing turnaround time. And better software tools are helping clinicians and researchers interpret the enormous volumes of genomic data generated.
That means NGS is no longer limited to elite research centers. It is becoming more practical for routine clinical and commercial use.
Diagnostics Is Emerging as One of the Strongest Segments
Among all applications, diagnostics is becoming one of the most exciting areas for NGS growth in the United States.
NGS-based diagnostics are increasingly used for:
Tumor profiling in oncology
Rare disease diagnosis
Prenatal and reproductive screening
Infectious disease detection
This is important because diagnostics is where sequencing moves from being “innovative” to being clinically essential.
In cancer care, for example, genomic testing is often needed to determine whether a patient is eligible for a targeted therapy or immunotherapy. In pediatric medicine, sequencing can uncover rare mutations that traditional diagnostic methods miss. In infectious disease management, genomic sequencing can support pathogen detection and surveillance.
As clinical confidence grows and physicians become more comfortable using molecular results in treatment planning, NGS diagnostics will likely become a standard part of care in more specialties.
Drug Discovery Is Becoming a Major Opportunity
Another major growth area is the use of NGS in drug discovery and pharmaceutical research.
Pharmaceutical and biotech companies are under intense pressure to develop drugs faster, reduce failure rates, and better identify which patients will benefit from treatment. NGS helps with all of that.
It is used for:
Target identification
Biomarker discovery
Pharmacogenomics
Companion diagnostic development
Clinical trial stratification
This makes sequencing valuable not just in the lab, but across the entire drug development lifecycle.
In a competitive pharmaceutical landscape, companies that can use genomic data to make better R&D decisions gain a serious advantage. That is one reason NGS is becoming deeply embedded in U.S. life sciences strategy.
The Services Segment Is Also Expanding
Not every company, lab, or hospital wants to buy expensive sequencing equipment and build a full internal genomics team. That is why the NGS services market is growing rapidly as well.
Sequencing service providers offer outsourced support for sample processing, sequencing runs, data interpretation, and clinical or research reporting. This model is especially attractive for organizations that need genomic capabilities but do not want to make a major upfront capital investment.
This trend is helping democratize access to NGS. Smaller institutions, emerging biotech firms, and specialized research teams can still participate in genomic innovation without owning every part of the infrastructure themselves.
But the Market Still Faces Real Challenges
Despite the strong growth outlook, the NGS market is not without obstacles.
One of the biggest challenges is data complexity.
Sequencing produces enormous volumes of genomic information, but generating the data is only part of the story. The harder part is interpreting it correctly. Many hospitals and laboratories still struggle with:
Data storage and management
Bioinformatics infrastructure
Clinical interpretation
Shortages of trained specialists such as bioinformaticians and genetic counselors
This creates a real bottleneck.
A second challenge is regulatory and reimbursement uncertainty. Not all sequencing-based tests are reimbursed consistently, and approval pathways can be complicated. For diagnostic labs and technology providers, this creates financial unpredictability and can slow market expansion.
So while the growth story is compelling, long-term success will depend not only on innovation but also on better integration, reimbursement clarity, and workforce development.
Sequencing by Synthesis Still Leads — But Competition Is Growing
From a technology standpoint, Sequencing by Synthesis (SBS) currently dominates the U.S. market. It remains the most widely used platform because of its high accuracy, strong scalability, and suitability for applications such as whole-genome sequencing, RNA sequencing, exome sequencing, and targeted panels.
However, alternative sequencing approaches are also gaining traction.
Ion semiconductor sequencing, for example, is becoming increasingly attractive in settings that require rapid turnaround and targeted analysis. Because it avoids some of the complexity of optical detection systems, it can be useful in smaller labs and certain clinical environments.
Over time, the market is likely to become more diversified as laboratories choose platforms based on speed, scale, application type, and cost efficiency.
Hospitals and Clinics Are Becoming a Key End User Group
One of the clearest signs that NGS is moving into the mainstream is the growing adoption among hospitals and clinics.
As genomic medicine becomes more relevant to oncology, prenatal care, inherited disease testing, and infectious disease management, healthcare providers are increasingly bringing sequencing closer to the point of care. Some are building internal capabilities, while others are partnering with outside providers.
This is a major market signal.
When hospitals start adopting a technology more widely, it usually means the technology is crossing the threshold from “advanced” to “necessary.” That is exactly what appears to be happening with NGS in the United States.
State-Level Innovation Is Strengthening National Growth
The U.S. NGS market is also being powered by strong regional ecosystems.
States such as California, New York, New Jersey, and Texas are playing a major role due to their concentration of biotech firms, pharmaceutical companies, medical institutions, research centers, and healthcare infrastructure.
California stands out because of its dense biotech and venture capital environment. New York benefits from its powerful academic and hospital networks. New Jersey remains important because of its pharmaceutical and life sciences footprint. Texas is emerging rapidly thanks to healthcare expansion, research investment, and a growing patient base.
This regional diversity is a strength. It means the U.S. market is not dependent on one city or one institution. Instead, it is being supported by a broad national ecosystem of innovation.
Final Thoughts
The United States Next-Generation Sequencing market is not just growing — it is becoming one of the most important pillars of future healthcare.
With the market projected to rise from US$ 4.62 billion in 2025 to US$ 20.23 billion by 2034, NGS is clearly moving beyond its role as a niche research tool and into the center of diagnostics, drug discovery, and personalized medicine.
The technology is becoming faster, more accessible, and more clinically relevant. Hospitals are adopting it. Pharmaceutical companies are relying on it. Researchers are scaling it. And patients are increasingly benefiting from the insights it can provide.
There are still hurdles to overcome, especially around reimbursement, data interpretation, and clinical integration. But the direction is unmistakable.






Comments
There are no comments for this story
Be the first to respond and start the conversation.